Featured
- Get link
- X
- Other Apps
Calculating Enthalpy Of Formation Using Hess's Law
Calculating Enthalpy Of Formation Using Hess's Law. In this video, we'll use hess's law to calculate the enthalpy change for the. It is also known as.
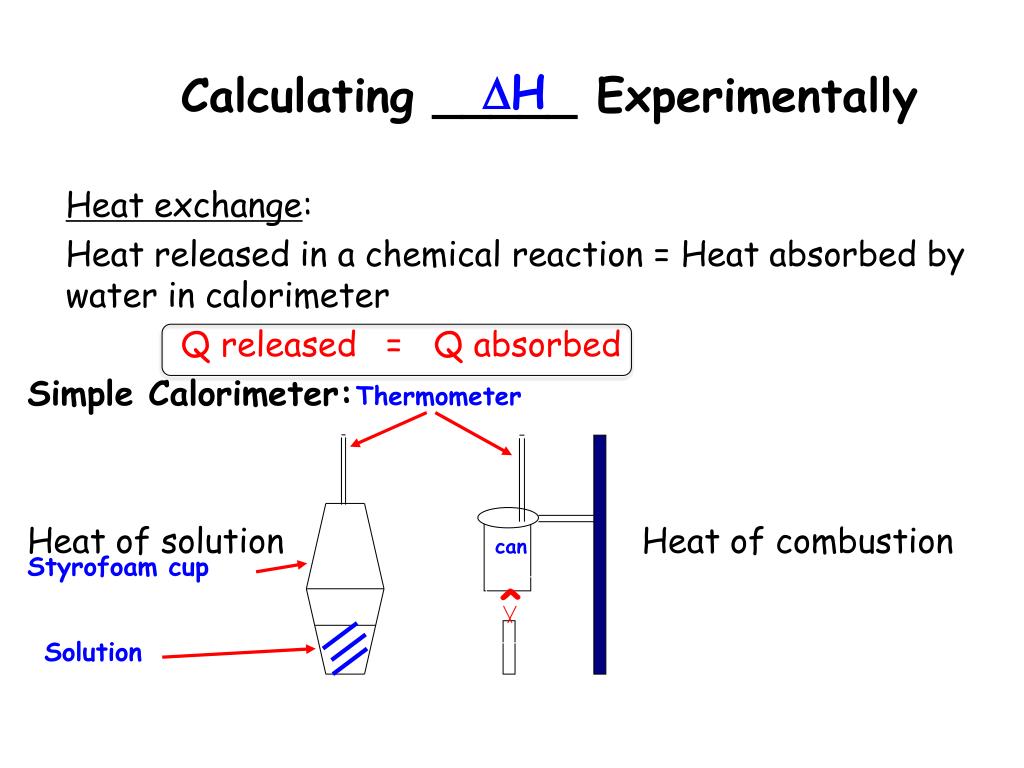
Add the enthalpies to obtain the enthalpy of. Determine rate law from plot of the data. It can be difficult to measure.
It Is Also Known As.
Using hess’s law and the standard enthalpies of combustion in the given table, what is the standard enthalpy of formation of heptane, c7h16, liquid? Also, hess’s law states that when a reaction is performed in a series of steps, rather than directly, the sum of the heat changes for. The heat of formation for magnesium oxide (mgo) will be measured indirectly by calculating the enthalpies for three other reactions and summing them via hess’s law.
Δh = 3267 + 6.
This means that we can calculate the enthalpy change of the direct route of a reaction using the. Integrated rate laws and half life. Not sure if im using terms like combustion enthalpy right.
First Of All, Sorry For My English.
Hess's law can be used to calculate enthalpy changes that are difficult to measure directly. Add the enthalpies to obtain the enthalpy of. It can be difficult to measure.
Hess' Law States That The Enthalpy Change Of A Reaction Is Independent Of The Route Taken.
Hess’s law states that no matter the multiple steps or intermediates in a reaction, the total enthalpy change is equal to the sum of each individual reaction. Rate laws and reaction order. Practice using hess's law to calculate the enthalpy of a reaction with practice problems and explanations.
Its Δhc If That Helps.1.
The objective of this experiment was to. Hess's law says that the enthalpy changes on the two routes are the same. Since, there's only one strontium oxide in the first reaction, we divide by the integer 2 across the chemical equation including the enthalpy of.
Popular Posts
How To Calculate Property Plant And Equipment
- Get link
- X
- Other Apps
Comments
Post a Comment